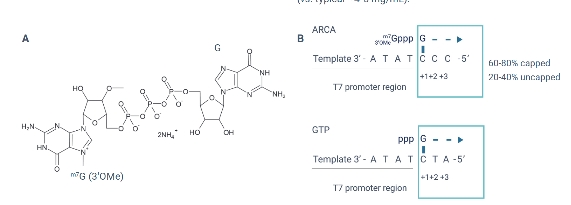
TriLink’s CleanCap® AU analog is designed for the co-transcriptional capping of RNA to produce self-replicating RNAs which are based on the genomes of positive sense (+) strand RNA viruses such as Venezuelan equine encephalitis virus (VEEV), Semliki forest virus (SFV), and Sindbis virus (SIN). The (+) strand genomes of these viruses start with a 5′-AU. Cap-1 RNAs produced by CleanCap AU are more suitable for eukaryotic usage than Cap-0 RNAs produced by legacy co-transcriptional capping methods such as anti-reverse cap analog (ARCA). Compared to enzymatic capping, CleanCap technology offers more streamlined and efficient manufacturing of self-amplifying RNAs. In addition, CleanCap AU analog can be used in conjunction with TriLink’s catalog of modified and unmodified NTPs.
Self-amplifying RNAs (also known as self-replicating RNAs), or saRNAs, are increasing in popularity as a platform for vaccine development. Serving as vectors, saRNAs use a viral backbone in which the viral structural genes are replaced with an antigen(s). Because they mimic a viral infection, saRNAs combine the benefits of live attenuated and subunit vaccines. They typically require lower doses than mRNA vaccines and are amenable to repeat dosing. The modular nature of saRNAs enables them to be rapidly deployed to novel disease targets. saRNAs may be particularly beneficial for personalized cancer vaccine development, which relies on a construct that can express multiple individualized neoantigens present in an individual patient’s tumor. Use of cell-free in vitro transcription (IVT) in making these vectors allows for more rapid and scalable manufacturing of saRNA vaccines than conventional ones.
Self-amplifying RNA replicons provide a promising platform for vaccine development. Alphavirus backbones, including Venezuelan equine encephalitis virus (VEE), Semliki Forest virus (SFV), and Sindbis virus (SIN), are frequently employed in construction of saRNAs. In the construct, the antigen gene replaces the viral structural genes, resulting in a non-infectious virus. Upon delivery, the saRNA replicates through a double-stranded intermediate, which increases its persistence and induces an innate immune response to promote antigenicity of the vaccine. The saRNA can be replicated in high copy inside the delivered cells, providing its potential for effectiveness even at low doses (e.g., in micrograms).
This article is posted at trilinkbiotech.com

Please fill out the form to access the content